Biology
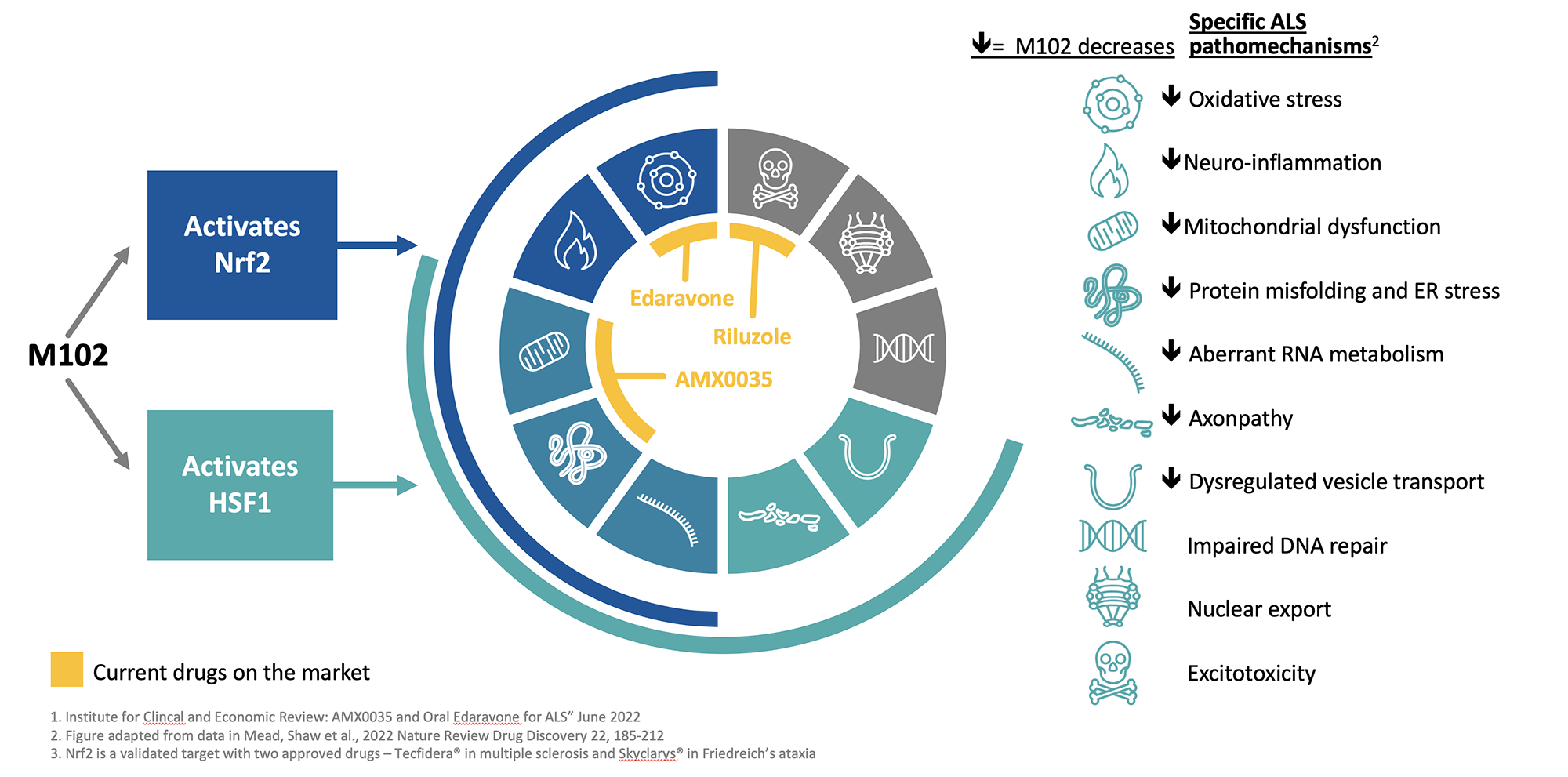
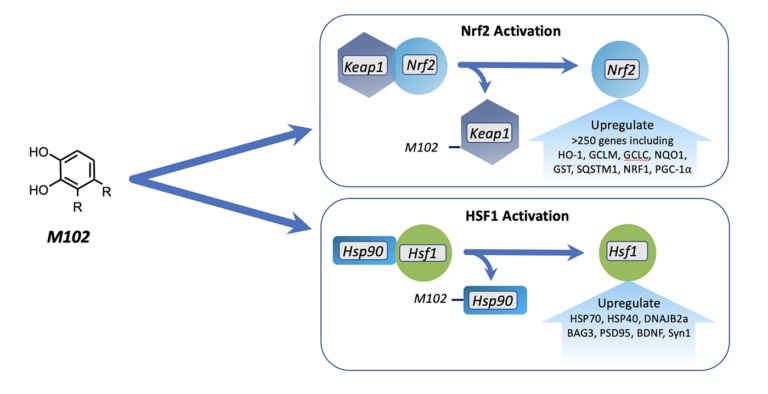
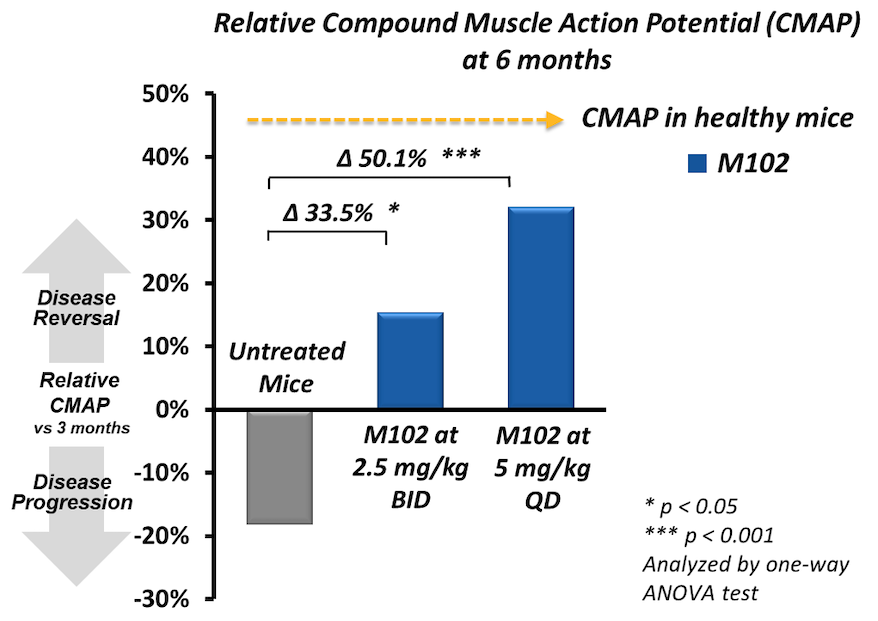
M102 is a dual activator of the NRF2 and HSF1 pathways which are disease-modifying pathways that are downregulated in ALS.
Through NRF2 and HSF1 activation, M102 reduces oxidative stress and inflammation, enhances mitochondrial biogenesis and initiates autophagy. In addition, M102 refolds misfolded proteins, disaggregates aggregated proteins, and clears misfolded and aggregated proteins.